Vesseloïds: A new model of guided self-organization allowing rapid formation of functional artificial vessels in one step
Domaine Santé et Bien-être
Technologie Ingénierie tissulaire et cellulaire
Challenges
Current methods of tissue engineering are limited to the production of non-vascularized reduced-size samples, dependent of the nutritional contribution of the culture medium.
Those processes prevent the vessels extraction for grafting and revascularization, which limits their use to in vitro studies.
Moreover, they do not allow to perfectly mimic the structure and histology of natural vessels, as they do not take into account the physiological context and stress forces that apply to the vessels.
The only method that is able to approach the properties of human vessels is a long and complex process.
There remains a need for a rapid process of formation of functional artificial vessels.
Innovative solution
One-step production of mature human tubular vessels from a microfiber composed of two cell types whose structure histology and mechanical properties are close to natural vessels.
APPLICATIONS
-
Tissue engineering: grafting of artificial vessels in engineered tissues to increase their viability with their vascularization
-
Pharmaceutical industries: drug screening, safety studies
COMPETITIVES ADVANTAGES
-
Functional, mature endothelial vessels
-
Generation in one step and one day of tailor-size small diameter artificial micro vessels with self-organization of the cells
-
Individualized and easy to handle, with the protective sheath of alginate that can be removed for grafting
-
Reproduction of the structure and histology of natural blood vessels
-
Reproduction of the physiological context and stress forces applied to the vessels
-
Perfusable, allowing the vascularization for the life-lengthening of engineered tissues
DEVELOPMENT STATUS: TRL 4/5
- Publication : L Andrique, G. Recher, K Alessandri, N. Pujol, M Feyeux, et al.. A model of guided cell self-organization for rapid and spontaneous formation of functional vessels. Science Advances, American Association for the Advancement of Science (AAAS), 2019.
- Until now, endothelial cells are HUVECs. In the near future, they will be derived from iPS.
How it works
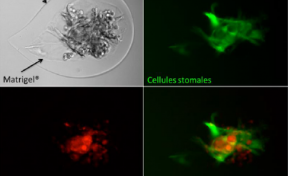
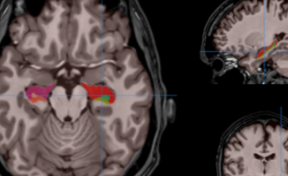
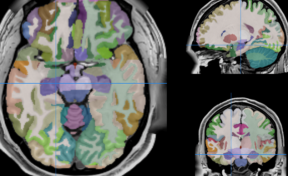
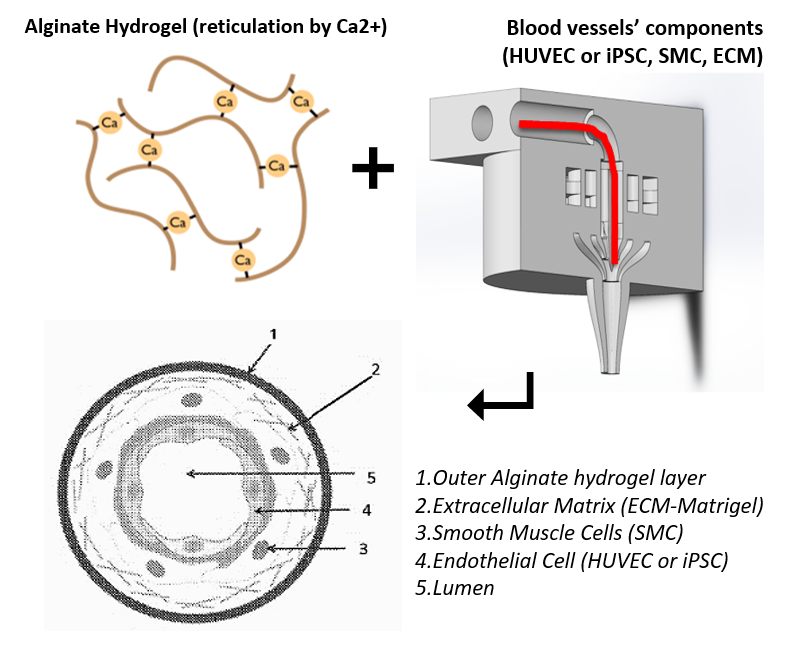
The inventors have developed a method of encapsulation of endothelial cells and smooth muscle cells in an alginate envelope, within which the cells organize themselves in homocentric layers around a lumen.
Production is done using a 3D-printed microfluidic coextrusion device, of long permeable hydrogel tubes in which Endothelial Cells and Smooth Muscle Cells are coencapsulated in the presence of an Extracellular Matrix CM.
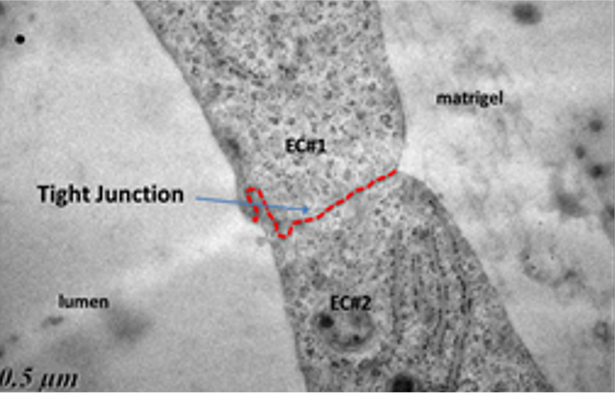
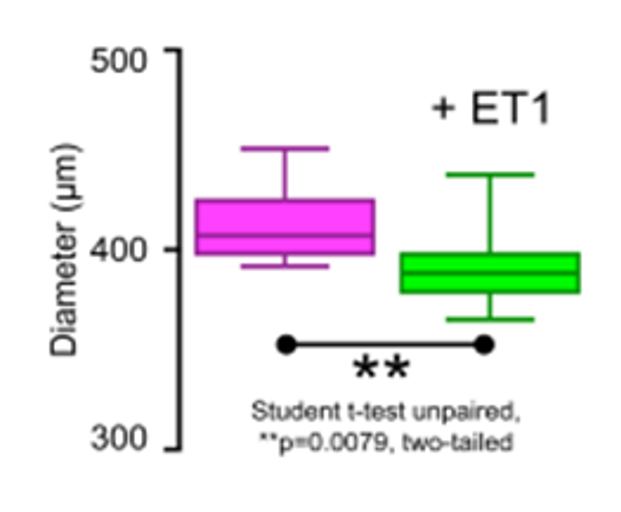
The produced microvessels (named “vesseloids” exhibit the main properties of mature blood vessels. In particular, (i) the typical histology of vessels is obtained within 1 day, (ii) the overall cell population becomes quiescent after a few days, and the vesseloids are (iii) perfusable and (iv) contractile in response to vasoconstrictor agents.
The outer layer of alginate hydrogel can strengthen the rigidity of the cellular microfiber and thus facilitate its handling. This layer can be removed for grafting.
CHARACTERISTICS
Generation of tailor-size small diameter artificial micro vessels from 250 to 450µm external diameters (potentially up to 1mm) and a lumen of 100 to 280µm respectively with self-organization of the cells in 24h of culture
 |
 |
 |
|
Manipulable &Perfusable |
Liquid-tight to 20 kDa |
Contraction by Angiotensin II application |
Inventeurs
Developed by two synergic laboratories with cross-cutting skills in microfluidic technology and angiogenesis :
- UMR 5298 “Photonics, Numerical and Nanosciences Laboratory”
- Inserm U1029 “Angiogenesis and Cancer Microenvironment Laboratory”
Propriété intellectuelle
Pending patents : FR, EP and US
PARTERSHIPS
License and/or R&D collaboration
Contact
Christophe ZABAWINSKI
%63%2e%7a%61%62%61%77%69%6e%73%6b%69%40%61%73%74%2d%69%6e%6e%6f%76%61%74%69%6f%6e%73%2e%63%6f%6d
+33 (0)6 66 86 20 25
-
Ref: #SBE_2017-002_202011